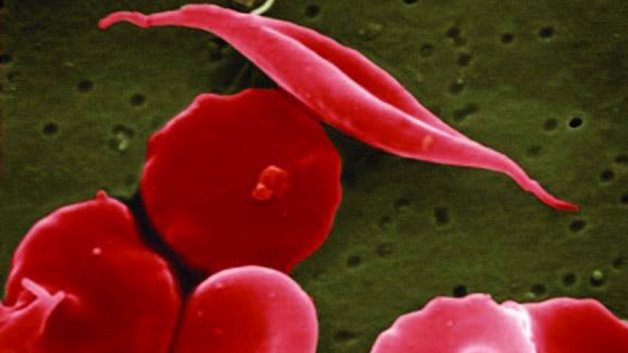
Regulators on Friday approved two gene therapies for sickle cell disease that doctors hope can cure the painful, inherited blood disorder that afflicts mostly Black people in the US.
The Food and Drug Administration said the one-time treatments can be used for patients 12 and older with severe forms of the disease. One, made by Vertex Pharmaceuticals and CRISPR Therapeutics, is the first approved therapy based on CRISPR, the gene editing tool that won its inventors the Nobel Prize in 2020. The other is made by Bluebird Bio and works differently.
The two gene therapies are the first approved in the US for sickle cell. The FDA has previously OK’d 15 gene therapies for other conditions. Some have list prices in the millions of dollars, and the sickle cell therapies will too.
In the U.S., an estimated 100,000 people have sickle cell and about a fifth of them have the severe form. Sickle cell is most common among Black people and 1 in 365 Black babies are born with the disease nationally. Scientists believe being a carrier of the sickle cell trait helps protect against severe malaria, so the disease occurs more often in mosquito-prone regions such as Africa or in people whose ancestors lived in those places.